Natural Product Development (lab 6d)
Purpose
We are performing this experiment to see if plants gathered from our field sites have useful antibacterial properties.
Materials
Balance, weigh boat, lab scoops
LB broth base
250 mL Media bottles
Sterilizer/autoclave
37 C. Water Bath
10 mL pipette
Plastic funnels
5 mm diameter Filter Paper disks
Beakers, 100 mL Syringe, 10 mL and filter
Reaction tubes and rack, 1.7 mL
Methanol, absolute
1 mL Pipette
Heat Block
Sterile LB Agar
Laminar flow hood and disinfectant
Bunsen burner and gas lighter
Inoculating loop, Ni/Cr wire
Sterile Petri dishes
E. coli JM109
Plant from field study site
Mortar and pestle
Fine-tipped Forceps
Ampicillin
Glass Spreader
37 C. Incubator Oven
Procedure
Part 2
We are performing this experiment to see if plants gathered from our field sites have useful antibacterial properties.
Materials
Balance, weigh boat, lab scoops
LB broth base
250 mL Media bottles
Sterilizer/autoclave
37 C. Water Bath
10 mL pipette
Plastic funnels
5 mm diameter Filter Paper disks
Beakers, 100 mL Syringe, 10 mL and filter
Reaction tubes and rack, 1.7 mL
Methanol, absolute
1 mL Pipette
Heat Block
Sterile LB Agar
Laminar flow hood and disinfectant
Bunsen burner and gas lighter
Inoculating loop, Ni/Cr wire
Sterile Petri dishes
E. coli JM109
Plant from field study site
Mortar and pestle
Fine-tipped Forceps
Ampicillin
Glass Spreader
37 C. Incubator Oven
Procedure
Part 2
- Grind 2g of plant using mortar and pestle with 10 mL of de-ionized water and wait three minutes.
- Filter through a paper-lined funnel
- Sterilize with filter and put into a 1 mL microtube
- Repeat w/ 10 mL Methanol
- Put tube in heat block with lid open and leave overnight
- Using sterilized forceps, insert 2 paper filter pieces in each tube
- Repeat with a positive control (Ampicillin) and negative control (Water) control tube
- Leave overnight at 4 degrees C.
- Use sterile pipette to transfer 1 mL of E. coli culture to petri disc (divided in 4 quadrants)
- Spread with flame sterilized glass spreader around disc, wait 15min. and cover.
- Use sterile forceps to place discs on plates in quadrants
- Incubate at 37 degrees C. for 24 H.
Results
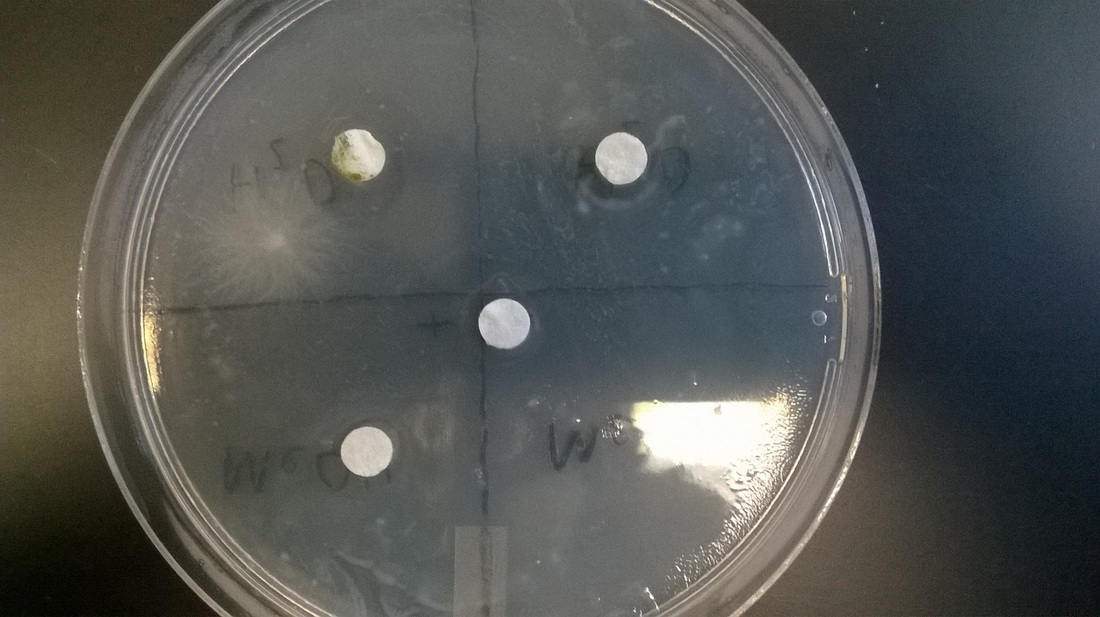
Both control disks performed as expected. The positive control produced a 1mm bacteria-free zone around it. The negative control was surrounded by bacteria, with a 1mm buffer between the bacteria and growth on the rest of the plate. Both extract disks showed no antibacterial properties. The methanol and water extracts produced a 1mm growth of bacteria around both.
Data Analysis
1. Did any extract give you positive results?
No, both extracts showed 1mm of bacteria growth around the disk.
2. Did your controls work as expected?
Yes, the negative control produced a 1mm ring of bacteria and the positive experienced little growth around the perimeter.
3. Discuss errors that could give you false results.
Our water extract disk also included the growth of some mold, which may have affected the bacteria growth. In addition, usual experimental error such as pipetting, cross contamination and misuse of filter paper could lead to false results.
4. Discuss further experimentation.
Further experiments could include different bacteria tests. In other words, this experiment could be repeated, substituting another harmful bacteria for e coli. This would show if the plant extract can counter any other harmful microorganisms.
5. Discuss next steps.
This experiment could be repeated with other plants from our field study sites. I know that my site contains several other types of plants and algae, some ow which could have antibacterial properties.
Conclusion
1. What did you like/find interesting?
I was surprised by the speed at which the bacteria was able to grow on our sample. Over the course of 24 hours, there was thick growth throughout the entire plate. I don't believe the conditions were ideal for growth either, as the petri dishes were left overnight in an unheated school building.
2. How did you and your partner collaborate?
For this lab, my partner was Peter Hunt, and because of this the lab was completed quickly and efficiently. We collaborated well and there were no major mishaps or mistakes.
3. What would you do differently next time?
The one thing that would need change was during the extraction process. Instead of adding the entire plant mixture to the filter, adding the liquid would suffice, as well as speed up the process and create less of a mess.
Thinking like a Biotechnician
1. If an extract gives a negative result in the antimicrobial assay, does that mean that the extract is not an antimicrobial agent?
No, this just means that the the plant extract is ineffective against that specific bacteria. While it may be likely that this plant may be ineffective against other bacteria, it may contain compounds that are harmless to one bacteria and fatal to another.
2. In preparing the sample disk, some of the methanol extractions smell like alcohol. Why is that a problem?
Alcohol is used to kill bacteria, which is why it was used in the positive control disk. If it is present in one of the extract disks, it will kill the bacteria regardless of any (if any) antibacterial properties of the extract itself. If that is the case, the results from that plate (or the entire experiment) are invalid.
3. Each extract may have one or more compounds in it. What should be done to begin to identify the exact compound in an extract that is causing the antimicrobial action?
Processes such as chromatography can be used to separate different compounds in a sample. After they have been separated, each compound can be replicated and tested (with experiments such as this one) individually to see if they hold antibacterial properties.
Both control disks performed as expected. The positive control produced a 1mm bacteria-free zone around it. The negative control was surrounded by bacteria, with a 1mm buffer between the bacteria and growth on the rest of the plate. Both extract disks showed no antibacterial properties. The methanol and water extracts produced a 1mm growth of bacteria around both.
Data Analysis
1. Did any extract give you positive results?
No, both extracts showed 1mm of bacteria growth around the disk.
2. Did your controls work as expected?
Yes, the negative control produced a 1mm ring of bacteria and the positive experienced little growth around the perimeter.
3. Discuss errors that could give you false results.
Our water extract disk also included the growth of some mold, which may have affected the bacteria growth. In addition, usual experimental error such as pipetting, cross contamination and misuse of filter paper could lead to false results.
4. Discuss further experimentation.
Further experiments could include different bacteria tests. In other words, this experiment could be repeated, substituting another harmful bacteria for e coli. This would show if the plant extract can counter any other harmful microorganisms.
5. Discuss next steps.
This experiment could be repeated with other plants from our field study sites. I know that my site contains several other types of plants and algae, some ow which could have antibacterial properties.
Conclusion
1. What did you like/find interesting?
I was surprised by the speed at which the bacteria was able to grow on our sample. Over the course of 24 hours, there was thick growth throughout the entire plate. I don't believe the conditions were ideal for growth either, as the petri dishes were left overnight in an unheated school building.
2. How did you and your partner collaborate?
For this lab, my partner was Peter Hunt, and because of this the lab was completed quickly and efficiently. We collaborated well and there were no major mishaps or mistakes.
3. What would you do differently next time?
The one thing that would need change was during the extraction process. Instead of adding the entire plant mixture to the filter, adding the liquid would suffice, as well as speed up the process and create less of a mess.
Thinking like a Biotechnician
1. If an extract gives a negative result in the antimicrobial assay, does that mean that the extract is not an antimicrobial agent?
No, this just means that the the plant extract is ineffective against that specific bacteria. While it may be likely that this plant may be ineffective against other bacteria, it may contain compounds that are harmless to one bacteria and fatal to another.
2. In preparing the sample disk, some of the methanol extractions smell like alcohol. Why is that a problem?
Alcohol is used to kill bacteria, which is why it was used in the positive control disk. If it is present in one of the extract disks, it will kill the bacteria regardless of any (if any) antibacterial properties of the extract itself. If that is the case, the results from that plate (or the entire experiment) are invalid.
3. Each extract may have one or more compounds in it. What should be done to begin to identify the exact compound in an extract that is causing the antimicrobial action?
Processes such as chromatography can be used to separate different compounds in a sample. After they have been separated, each compound can be replicated and tested (with experiments such as this one) individually to see if they hold antibacterial properties.